Introduction
Tisagenlecleucel (tisa-cel) is a second generation, CD19-targeted Chimeric Antigen Receptor (CAR) T-cell therapy for relapsed or refractory (R/R) large B-cell lymphoma (LBCL). The pivotal phase 2 trial JULIET included 93 patients and reported an overall response rate (ORR) of 52% and a complete response rate of 40% among treated patients. However, very little is known regarding its safety and efficacy in the commercial or real-world setting as well as from an intention-to-treat perspective. In our study, we report clinical outcomes of patients with R/R LBCL treated with commercial tisa-cel in 10 Spanish institutions.
Methods
Data were collected retrospectively from all consecutive patients with R/R LBCL who underwent apheresis for tisa-cel at a Spanish site from December 2018 until June 2020. Evaluable patients included those who received a CAR-T infusion and had at least 1 month of follow-up. Adverse events after infusion were graded according to the ASTCT consensus and efficacy outcomes were assessed according to the Lugano criteria. Efficacy outcomes were calculated in the patients who received a CAR T-cell infusion and in all patients who underwent apheresis for tisa-cel (intention-to-treat).
Results
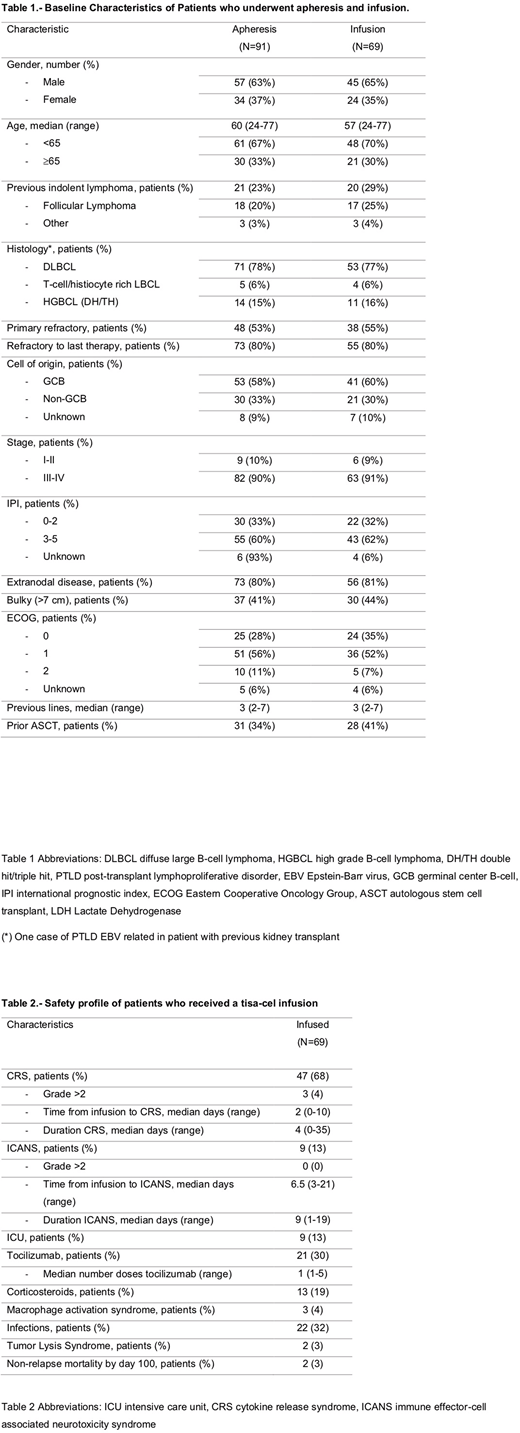
During the study period 91 patients with R/R LBCL underwent apheresis for tisa-cel. At the study cutoff, 69 (76%) patients had received a CAR T-cell infusion whereas 22 (24%) had not due to progressive disease (n=10, 45%), pending manufacturing process (n=7, 32%), out of specification (OOS) or manufacturing failure (n=4, 18%), or others (n=1, 5%). Sixty-one patients had at least the first disease response evaluation at 1-month post-infusion. Baseline characteristics of the whole cohort and the infused patients are summarized in Table 1. Among infused patients, median age was 57 years (range 24-77) and 65% were male. Most of the patients had a high-risk International Prognostic Index score (62%), an advanced stage at diagnosis (91%) and were refractory to the previous therapy (80%). Median follow-up after CAR T-cell infusion was 4 months (1-16).
Fifty-six patients (81%) received bridging therapy before infusion, including chemotherapy in most cases (n=51, 91%). All patients received fludarabine and cyclophosphamide as lymphodepleting chemotherapy. Median time from apheresis to infusion was 53 days (range 29-225). Median infused cell dose was 2.2 x 108 CAR positive viable T-cells (range 0.4-4.2 x 108). Eleven (16%) products were considered OOS although 45% of them were infused.
Among the infused patients, 47 (68%) and 9 (13%) developed any grade of cytokine release syndrome (CRS) and neurotoxicity, respectively. Grade >2 CRS and neurotoxicity events occurred in 3 (4%) and 0 patients, respectively. Tocilizumab was administered to 21 (30%) patients and steroids to 13 (19%) patients. Nine (13%) patients required admission to the Intensive Care Unit. By day 100, 2 (3%) patients experienced non-relapse mortality. Other adverse events including infections and tumor lysis syndrome are summarized in table 2.
Best response achieved among the infused patients included complete remission in 17 (28%) patients and partial remission in 22 (36%) patients, with an ORR of 64%. Stable disease and progressive disease were the best response in 4 (7%) and 18 (29%) patients, respectively. Considering all patients who underwent apheresis, CR and PR were obtained in 19% and 24% (ORR 43%), respectively.
Median progression-free survival (PFS) and overall survival (OS) for infused patients were 3 months (95%CI 2.1-3.4) and 8 months (95%CI 5.7-10.8), respectively. In the univariate analysis, patients with ECOG score >1 (HR 4.3 95%CI 1.5 - 40.9) (p < 0.001) and IPI score >2 (HR 4.8 95%CI 1.6 - 14.4) (p = 0.002) were associated with lower PFS while an ECOG score >1 was also associated with lower OS (HR 3.4 95%CI 1.15 - 34.4) (p = 0.04). In the intention-to-treat analysis for all patients who underwent apheresis, median PFS and OS from apheresis were 4 months (95%CI 2.8-5.8) and 9 months (95%CI 5.8-11.8), respectively.
Conclusions
Treatment with tisa-cel was able to induce disease response with a good toxicity profile in a population of patients with LBCL treated in a European country. Poor performance status at the time of CAR T-cell infusion was associated with lower PFS and OS.
Iacoboni:Novartis, Gilead, Celgene, Roche: Honoraria. Guerreiro:Novartis: Honoraria. Mussetti:Novartis, Gilead: Honoraria, Research Funding. Sancho:Roche, Janssen, Celgene, Novartis, Gilead, Sandoz, Mundipharma y Takeda; Advisory y/o consultor para Roche, Janssen, Celgene, Novartis, Gilead, Sandoz, Celltrion y Kern-Pharma.: Honoraria. Hernani:Gilead: Honoraria. Sureda Balari:Takeda: Consultancy, Honoraria, Speakers Bureau; Celgene/Bristol-Myers Squibb: Consultancy, Honoraria; Merck Sharpe and Dohme: Consultancy, Honoraria, Speakers Bureau; Sanofi: Consultancy, Honoraria; Novartis: Consultancy, Honoraria; Gilead/Kite: Consultancy, Honoraria; Janssen: Consultancy, Honoraria; Incyte: Consultancy; Celgene: Consultancy, Honoraria; BMS: Speakers Bureau; Roche: Honoraria. Martin Garcia-Sancho:Celgene, Eusa Pharma, Gilead, iQuone, Kyowa Kirin, Roche, Morphosys: Consultancy; Roche, Celgene, Janssen, Servier, Gilead: Honoraria. Kwon:Jazz: Consultancy, Honoraria; Gilead: Consultancy, Honoraria; Novartis: Consultancy, Honoraria; Pfizer: Consultancy, Honoraria. Reguera:Celgene, Novartis: Consultancy; Novartis, Gilead: Honoraria. Barba:Novartis, Celgene, Gilead, Pfizer, Amgen, Roche: Honoraria.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal